#Iso 13485 consultants iso#
The organizations which have undergone or achieved ISO 13485 Certification Bodies in Paraguay have improved in their products quality, regulatory compliance, reliability and are in relation with the industry best practices. ISO 13485 Certification Consultants in Paraguay acts as a standard for The industries that take part in medical manufacturing.
#Iso 13485 consultants verification#
Verification of both input and output must be prepared before designing the process.Īdvantages of ISO 13485 Certification in Paraguay: The exhibition has to be achieved with environmentally friendly components and materials together with ensuring that the facility is secure to make sure all of the adequate activities. Once the design Is ready it is very important to examine it in front of the customer before going with the meeting. Manufacturing procedures and other vital specifications concerning surroundings have to be understood before demonstrating the procedure. Inspection has to be done before conducting any experiments on the meeting. Identification of the product and knowing the manufacturing companies. The output consists of raw materials particles that are required to fulfill the entire product.

The outcome of This input has to be decided with the assistance of the management system.

Considering all the appropriate study producing a lawful and statutory requirement in front of the management system can help to reduce the best design. The study of present market values concerning medical instruments and other costing matters must be discussed among the best management staff. Risk control and RISC addressing techniques implemented in the management. Some of the expectations in the clients in the medical industry are easily achieved with the help of the ISO 13485 Consultants in Paraguay. The design must Be of equality where it must fulfill the customer requirement. Allocating time and managing risk activities to be able to complete the entire project will help to finish the stage. This particular phase aids the organization in identifying aims and objectives. When the plan is Ready the next stage starts with the layout and development of a management strategy program. Preparation of Policies during the initial stages to make a nice structure and infrastructure of this organization will hold roles and obligations in the top management system.Įstablishment Activities to cut down the best strategy is the most important crucial measure of execution of any International standard.
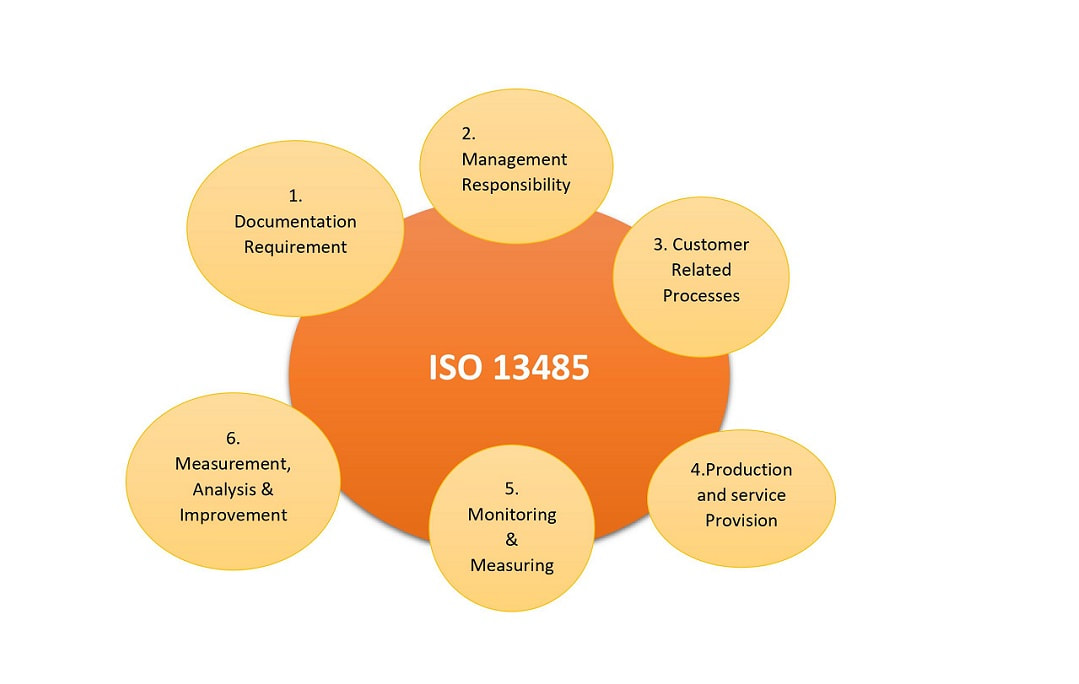
Also, about the specific requirement from the customers, the organization will have the ability to fulfill the customer needs with a proper medical device management system. Implementation of ISO 13485 Certification in Paraguay within an organization will assist the management system to design and develop a process for the medical devices. Medical industries must go through many deals to be able to generate a regulation, security product, and a safe item. Laying down a Strategy to produce a design is a really basic step to come up with a thriving medical device process.
#Iso 13485 consultants how to#
How to design and develop ISO 13485 standard in an organization?
